
Using the molar mass of a substance, one can find out how many moles are present in a sample of that substance. Molar Mass of O2 and Other Molecules Importance A mole of water, for example, has a molecular weight of 18.015 atomic mass units, and weighs approximately 18.015 grams. The relationship is as follows:ġ mole of H2O = 2 × 6.02214076 × 10 23 of Hydrogen + 6.02214076 × 10 23 of Oxygen.Īccordingly, the mass of one mole of a substance is equal to its molecular weight. One molecule of water is composed of one oxygen atom and two hydrogen atoms, so one mole of water is composed of one oxygen atom and two hydrogen atoms. Here is an example of how moles simplify chemical representation and calculations. Avogadro’s number can be used to understand how chemicals interact and how molecules within chemical samples interact. In calculations that involve more than one chemical, Avogadro’s number and moles must be used. It is done so that weight can be compared to the number of atoms within the substance (as defined by the number of moles) since weight is easier to measure than the number of atoms in the chemical sample. By representing 6.02214076 × 10 23 atoms, molecules are used in chemical calculations. 
The chemical reactions involve billions of atoms interacting with one another and being rearranged, but the movement of billions of atoms would be impossible to represent.Įven so, scientists still need a unit of measurement that can represent billions of elementary entities. In chemistry, Avogadro’s number and the mole are important concepts. Therefore, the number of atoms is 6.02214076 × 10 23, also known as Avogadro’s number. One mole of carbon 12 contains the same number of elementary entities as there are atoms. Mole counts are used by scientists to determine the number of elementary entities present in a chemical sample. A mole is a unit of measurement used to describe the quantity of a substance. In the case of moles, this definition isn’t very useful. The molar mass of a chemical substance is the mass contained in one mole of that substance, the mass of one mole of a given substance. Add the masses of all the elements in the molecule to find the molecule’s mass. Multiply the atomic mass of an element by the number of atoms in the molecule to find the molecule’s mass. The molecular mass of an element is equal to the sum of the masses of its constituent elements. The formula mass of calcium hydrogen carbonate is 117.10 amu, and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). We will use the term molar mass if we are discussing a mole of an ionic compound. Ionic compounds do not have individual molecules. When referring to compounds that are not molecular (ionic compounds), the term “molecular mass” is improper, and “formula mass” is generally used instead. Consequently, N2 has a molar mass of 28.02 grams per mole. 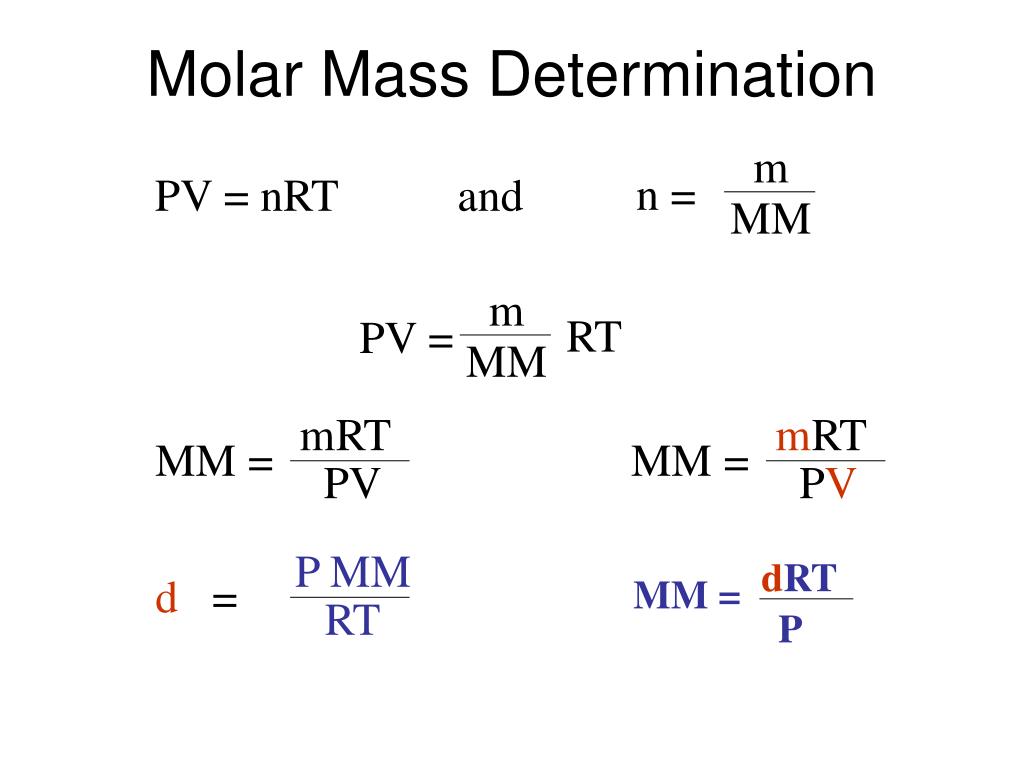
The molecular mass of a molecule is the result. The atomic mass of an element is the sum of the atomic masses of all its constituent atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
